Reading time: 12 minutes
Author: Thomas Masuch
While orthopedic technology is not mentioned explicitly in most market analyses, except as part of the higher-level Medical/Healthcare category, it has slowly developed into one of the most important user industries for Additive Manufacturing. According to market experts, this is thanks to years of consistently high growth. For most orthopedic and prosthetic products, AM is even today able to replace or at least complement traditional manufacturing processes. The increasingly widespread adoption of digital manufacturing processes offers opportunities for manufacturers along the entire AM process chain, from 3D scanning and CAD software to 3D printing and post-processing, and for AM service providers in particular.

Despite the lack of available data, the successful development can be measured by numerous indicators: “25 to 30 percent of our qualified leads at Formnext 2025 came from the O&P (Orthopedics & Prosthetics) sector,” reports Emil Wörgötter, who has been working as an AM engineer in orthopedic technology for almost ten years and is an application consultant at DyeMansion with responsibility for the EMEA and APAC regions. Among the more than 1,300 systems in active use at DyeMansion, a similar percentage is used for manufacturing O&P products, according to Wörgötter’s estimate. A definitive statement is difficult, however, as much of the business is conducted via service providers. The importance of the orthopedics market for the provider of end-to-end post-processing workflows (which encompass depowdering and finishing to the dyeing of 3D-printed plastic parts) is also evident from the fact that two of DyeMansion’s application consultants, Emilie Simpson (USA) and Emil Wörgötter (EMEA & APAC), specialize in the O&P market.
Market and growth
The stable growth of AM in orthopedics is the result of a constantly growing overall market (driven, in part, by an aging society in many industrialized countries) and the ever-increasing use of AM by the industry. Between 2008 and 2024, according to figures from the National Association of Statutory Health Insurance Funds (GKV-Spitzenverband), expenditure by statutory health insurers on medical aids such as orthoses and prostheses as well as glasses and hearing aids, etc. increased by 105 percent to € 11.7 billion.
Various orthoses printed by Nowecor Images: Emil Wörgötter
This development is being accelerated by a continued decline in the capital expenditure required to set up professional digital manufacturing processes and get started with Additive Manufacturing. Depending on system size, purchase prices for some powder bed 3D printing systems (for polymer materials) are now in the five-digit range. Together with increasingly affordable 3D scanners, an entry-level AM production line can now be set up for less than € 100,000, says Wörgötter. DyeMansion is also playing its part by offering the VX1, a chemical smoothing system, in the lower price range. Wörgötter expects further dynamic application growth, in particular due to the increasing automation and simplification of design processes. According to him, this represents the biggest hurdle on the horizon. He sees opportunities for orthopedic technology companies and AM service providers as well as some urgency to get involved: “AM in this industry really is in the fast lane. If you don’t get on board now, you risk being left behind.” The success of those who are already successfully working digitally is the result of the work of recent years. “Now is a good time to act, because the solutions are mature, but the market is still catching up.”
There are around 5,300 medical supply stores in Germany, spread across just under 2,200 owners, 1,465 of whom are sole entrepreneurs with only one location. Every year, these supply the population with almost 25 million medical aids, according to data provider PM Pflegemarkt.com. Emil Wörgötter estimates that in the DACH region (Germany, Austria, Switzerland), around 10 to 20 percent of orthopedic technology companies have adopted digital technology to some extent, i.e., a 3D scanner at the very least. “This is, in a sense, the ticket to the digital world.” Design and manufacturing can also be outsourced if necessary, so that not “all steps have to be taken at once,” thus avoiding overburdening the company and its employees. AM service providers or partners such as the Nowecor AG purchasing association (see report ) can help with this. Only a small proportion manufacture printed, ready-to-deliver “definitive supplies” themselves; examples include innovative, family-run orthopedic companies such as Kriwat (see article ) or large companies such as Otto Bock.
Cranial helmet, which was presented by DyeMansion at Formnext 2025. Image: Thomas Masuch
Still plenty of potential
Even though the industry has grown steadily in recent years, Wörgötter estimates that less than 5 percent of orthotic and prosthetic devices are currently 3D printed. Extrapolated to the total number of devices, this is already a considerable number of products in Germany alone, but it also shows that there is still great, untapped market potential. Wörgötter expects the share of 3D-printed orthopedic products to increase significantly in the future: “We are in a phase where pragmatists are starting to follow the tech-savvy early adopters in jumping on the bandwagon because the advantages are so obvious. The proportion of 3D-printed products on the market will therefore surely rise. The big question is when.” Precise predictions are difficult here, as market dynamics and health policy decisions play a big part.
To further advance and accelerate the development of Additive Manufacturing in the field of orthopedics, DyeMansion works closely with all 3D printer manufacturers in the polymer powder bed sector to offer complete solutions. With HP, for example, it produces industrial, automated solutions for large companies in the O&P sector. Both companies organize seminars and workshops on digitalization in orthopedic technology. One success story is end user Orfi (orthopedic shoe technology company from Mörfelden-Walldorf, Germany), which has many of its products and components 3D-manufactured by DyeMansion partner Nowecor AG.
International differences
DyeMansion has been focusing on the orthopedic sector for years and, in 2021, supported Otto Bock with FDA 510(k) clearance for the Mycro Band cranial helmet (manufactured by HP MJF from PA12 with DyeMansion’s Deepdye Coloring). DyeMansion technologies are also used in the finishing of the Sprout3D cranial helmet from Surestep (a Hanger Corporation company).
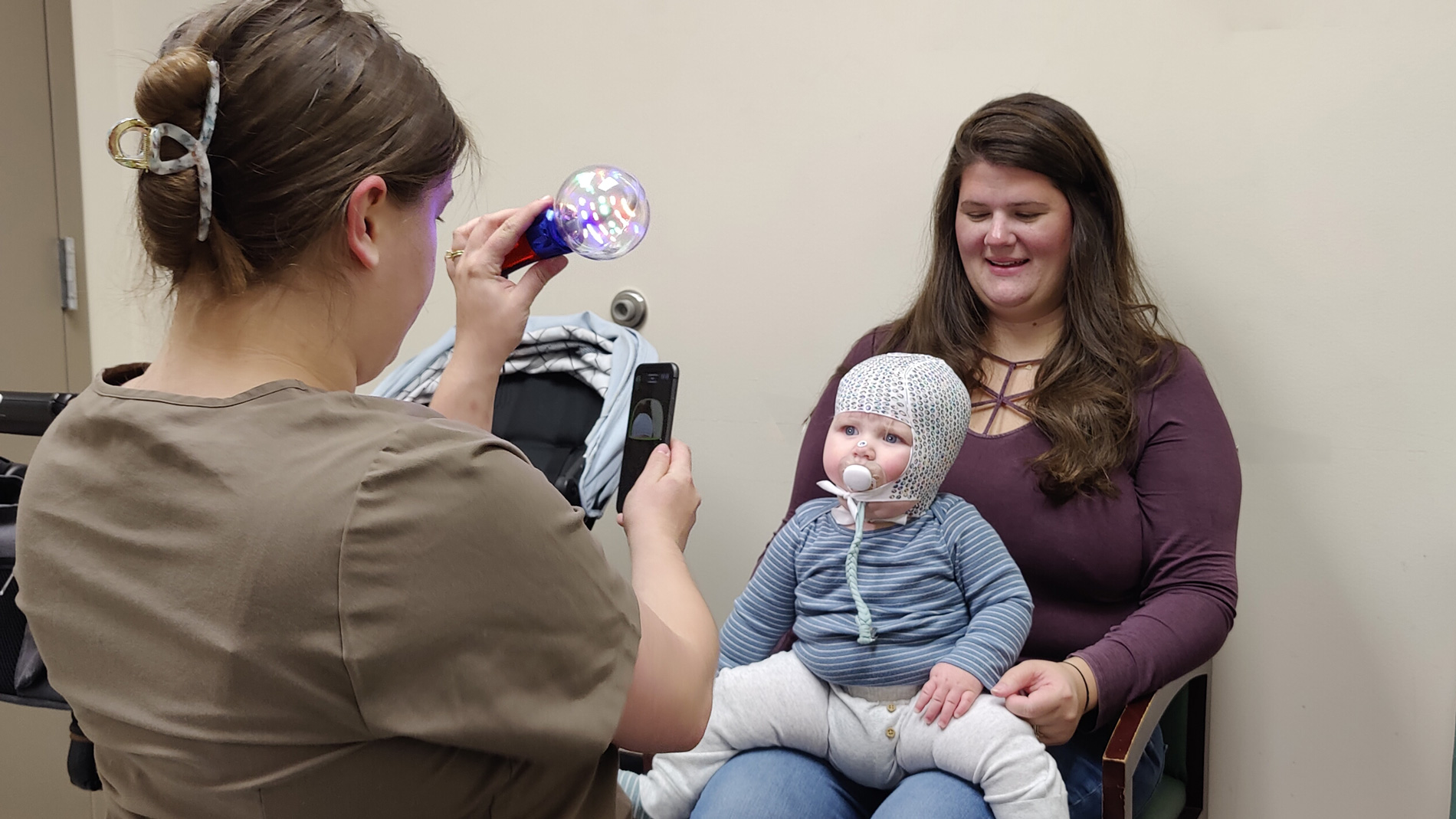
Such cranial helmets are used to correct head deformities in infants, either for medical or aesthetic reasons. In Germany, the costs for aesthetic treatments are not usually covered by health insurance. In the US, too, such helmets often have to be paid for privately. However, according to Wörgötter, it is more common for people in the US to pay for medical services out of their own pockets, which is why such helmets are in high demand there.
Scanning process for a cranial helmet at Hanger Clinic Prosthetics & Orthotics in Baltimore (USA) for taking patient-specific measurements for the manufacture of custom cranial orthoses. Image: Lester Hitch
Added value is key
Paying orthopedic products out of the patient’s pockets leads to greater competition and pressure to innovate. “Suppliers must offer patients and their parents real added value.” The advantages of 3D-printed cranial helmets are significant and range from more precise pressure distribution on the head to lower weight and better ventilation to more comfortable care not only for children but also for their parents (scanning eliminates the need for plaster casts).
This development has led to cranial helmets increasingly being produced on 3D printers in the US. “We are already seeing a trend here toward additive manufacturing replacing conventional manufacturing,” says Wörgötter. This is also due to another special feature of the US orthopedic market: Care in the US is primarily determined by large companies. Global player Hanger, for example, has an estimated market share of around 20 percent. “And these large companies are also more willing to roll out digital manufacturing processes than small businesses with a tradition of craftsmanship, such as those in Germany.”

Focus on efficiency
In Germany, DAFO (dynamic ankle foot orthosis) is a device for which many companies already use 3D Printing. This is partly because it is one of the most common medical devices to custom make, and partly because it fits into smaller printer build spaces and is easy to “pack.” Since such products are usually covered by health insurance, Wörgötter says that, in addition to the added value of the product, specialist companies are primarily focused on increased efficiency and standardization in the processing of care services. Here, too, 3D printing can offer a real advantage: Digital workflows relieve the few available specialists by eliminating simple production steps such as pouring plaster and allowing the orthotic shells to be produced overnight in the printer instead of having to be deep drawn by hand. Personnel costs for production are reduced, and specialists can devote more time to delivering added value to the patient. “This is a particularly important factor in view of the shortage of skilled workers.”
And because the investment required for Additive Manufacturing as a whole poses greater challenges for a small specialist company than for global players such as Össur or Otto Bock, the majority of supplies in Germany are ordered through service providers. “Even though most of the service providers do not specialize exclusively in orthopedic products, their expertise in this area is growing,” says Wörgötter. Service providers also offer certificates confirming the biocompatibility of the materials used and post-processing steps. This is a special responsibility that DyeMansion also honors.